[050] Cellular respiration (GB#101H01) | 基礎医学教育研究会(KIKKEN)Lab

● Cells that absorbed oxygen exhale carbon dioxide
The task of extracting energy from nutrients absorbed in the gastrointestinal tract is responsible for each cell making the body. To turn its work, the cell takes oxygen and exhales carbonic acid gas.
—
Contents
- 1 ● Response of respiration
- 2 ● Oxygen and carbon dioxide enter and leave by diffusion
- 3 ● ATP per cell (calculation redo)
- 4 ● Oxygen and carbon dioxide per cell
- 5 ● What about cultured cells?
- 6 ● Red blood cells do not consume oxygen
- 7 ● Oxygen overflows from capillaries
- 8 ● How much surplus oxygen can afford
- 9 ● Carbon dioxide gas that is discharged is also carried by the blood vessel
- 10 ○ Referenced sites
- 11 ○ Related articles
- 12 ○ Referenced books
● Response of respiration
The nutrients of general cells, energy sources are mainly glucose and fatty acids. In glucose and fatty acid the structure and properties of the molecule are completely different. However, both are compounds of carbon, oxygen and hydrogen. In the process of decomposing this energy is gradually taken out to another molecule, and finally (although it is not really the end though) the energy is subdivided into many ATP (Adenosine Tri Phosphate).
ATP is said to be also called “intracellular energy currency” because the energy incorporated in ATP can be used in a variety of different applications, basically in a common way in the cell. Nutrients originally made from carbon, oxygen and hydrogen, after all the biologically available energy is extracted, eventually become carbon dioxide and water molecules and finish their functions.
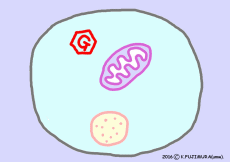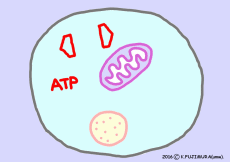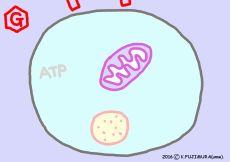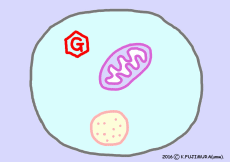
The work of extracting the energy that the nutrient has to the end is carried out by the reaction of citric acid cycle (= TCA cycle) in mitochondria. However, before entering the citric acid cycle, pretreatment according to the material must be carried out. ![]()
If the material is glucose, it turns into two pyruvic acid by glycolysis reaction outside mitochondria and then enters the citric acid cycle via acetyl-CoA in mitochondria.
In the case of fatty acids, after entering the mitochondria, they undergo a process called beta-oxidation, and then enter the citric acid cycle via acetyl-CoA.
Even if oxygen is not taken as occasion, only glycolysis from glucose proceeds. However, the process in the mitochondria does not operate when oxygen gas does not enter.
It is expressed as, in the case of glucose(C6H12O6),
C6H12O6 + 6H2O + 6O2 → 6CO2 + 12H2O + Energy(686 kcal/mol・glucose)
Or, by rearranging the number of water molecules before and after the reaction,
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy(686 kcal/mol・glucose)
When extracting energy only with glycolysis in the absence of oxygen, mitochondria is closed and no carbon dioxide is emitted. Only two ATPs are formed in the cytosol, pyruvic acid is converted to lactic acid, and taking out energy from this molecule is a rest for now.
When oxygen is sufficient and all energy available from one glucose molecule is extracted out, approximately 30 molecules (38 molecules in old textbooks) of ATP are made. The energy released from 1 mole (6.02 × 10 23 molecules) ATP is 7.3 kcal (standard state). If it is 30 mol, the energy is 219 kcal, only one third of 686 kcal. Still it is said that the recovery rate of energy is higher than that of artificial institutions such as gasoline engines, as it extracts energy from organic matter. In the cell, energy that could not be transferred to ATP is also used as heat to maintain body temperature, so there is no waste in energy usage.
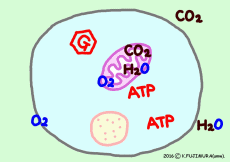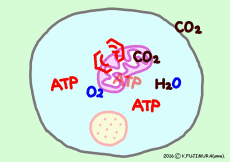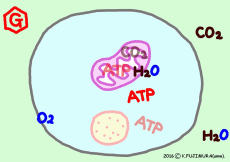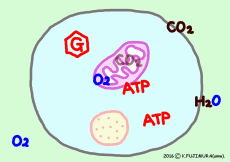
● Oxygen and carbon dioxide enter and leave by diffusion
Gases such as oxygen and carbon dioxide pass through by diffusing the cell membrane. It expands from the high concentration side to the low concentration side, from the higher partial pressure side to the lower side. Since oxygen is consumed in the mitochondria one after the other, it moves from the outside of the cell, carbon dioxide moves naturally out of the cell as it occurs from mitochondria one after another.
Glucose, on the other hand, can not diffuse cell membranes arbitrarily. A glucose transporter, which is a dedicated carriage protein buried in the cell membrane, is carried from the higher concentration of glucose to the lower side. When glucose is used more and more in the cell, it will be carried from outside the cell. On the other hand, fatty acids pass through the cell membrane by diffusion.
Energy-transferred ATP is basically used only in the cell, not outside. Special transport protein is prepared for lactic acid, and it seems that entry and exit are regulated as necessary.
● ATP per cell (calculation redo)
“There is always about 109 (1 billion) molecules of ATP in each cell, and in many cells all ATPs are consumed and replaced every 1-2 minutes” (Essential Cell biology, 2010).
The minimum amount of energy (basal metabolic rate, BMR) consumed by humans only in one day is about 1500 kcal for adults weighing 60 kg.
It will be good if you live normally, it will be about 2,300 kcal. Since 30 moles of ATP is made from 686 kcal of energy of 1 mol of glucose, it is estimated that ATP produced on the human body on a daily basis in this state is 30 × 2300/686 ⇒ 100 mol.
ATP produced (= consumed) per minute is 100 / (60 × 24) ⇒ 100/1440 ⇒ 0.0694 = 6.94 × 10 -2 mol (ATP) /min.
Recent data estimate the total number of cells of adult human with a body weight of 70 kg is 37 trillion (3.7 x 10 13) (it is said that roughly 60 trillion pieces are rough it was …). If it is 60 kg, it becomes 3.7 × 60/70 × 10 13 ⇒ approximately 3.2 × 10 13. By dividing the amount of ATP produced in the whole human body by the number of cells and calculating the ATP consumption per minute of one cell, 0.0694 / 3.2 × 10 -13 ⇒ 0.0217 × 10 -13 ⇒ 2.17 × 10 -15 mol /min/cell.
From now on to the number of molecules, it becomes 2.17 × 10 -15 × 6.02 × 10 23 ⇒ 2.17 × 6.02 × 10 8 ⇒
· 13.1 × 10 8 molecule (ATP) /min/cell (about 1.3 billion molecules of ATP)
About 10 9 (1 billion) molecules. It is almost within the allowable range, isn’t it?
● Oxygen and carbon dioxide per cell
If this is all glucose as an energy source, 30 molecules of ATP are produced per glucose molecule, so the glucose required for 1 minute is 13.1 / 30 × 10 8 molecule/min. In fact, fatty acids are more useful as energy sources, but glucose is easier to calculate. Six molecules of oxygen are used to extract all energy from one glucose molecule. Therefore, the number of molecules of oxygen required per minute per cell is 6 times the number of glucose molecules, 6 × 13.1 / 30 × 10 8 molecule ⇒
· 2.61 × 10 8 molecule (O2) /min/cell, (about 260 million molecular oxygen)
In the case of glucose, the amount of carbon dioxide exhaled is the same as consumed oxygen, so the value CO2 / O2 (respiration quotient) is 1.0. When another nutrient or fatty acid is used as an energy source, the proportion of oxygen atoms in the molecule is much smaller than that of glucose, so that on average an oxygen consumption is increased by about 40% compared with the carbon dioxide discharged. Therefore, the breathing quotient is about 0.7. The average breathing quotient of actual breathing of the whole body seems to be about 0.8.
● What about cultured cells?
In cell culture, glucose is often given mainly as an energy source. Then, the oxygen consumed and the discharged carbon dioxide are 1: 1. In the case of cultured cells of the liver, if 1 x 10 5 cells per sheet 1 cm2 of the cell population grown without voids, 40 pmol (O2) /s/cm2 oxygen is required. Since it is 60 times in one minute, it becomes 2400 pmol /min/cm2.
Calculating from that, the number of oxygen molecules required per minute in this cell population is 2400 pmol = 2400 x 10 -12 mol = 2.4 x 10 -9 mol ⇒ 6.02 × 10 23 × 2.4 × 10 -9 molecule ⇒ 6.02 × 2.4 × 10 14 molecule ⇒ 14.5 × 10 14 molecule ⇒ 1.45 × 10 15 molecule (O2) /min/cm2. For each cell, 145 × 10 13 / 10 5 ⇒
・ 145 × 10 8 molecules (O2)/min/cell (14.5 billion molecular oxygen)
It is roughly 50 times the average oxygen consumption per cell calculated from ATP consumption of the whole body.
In another cell example, there is data that approximately 0.12 pmol of oxygen per hour is required for one cell of myocardial fibroblasts (cardiac fibroblasts). It has less metabolism than the liver. However, since it is a muscle cell, the metabolism will be bigger than the non-moving cell. 0.12 × 10 -12 mol/h ⇒ 120 × 10 -15 mol/h ⇒ 120 / 60 × 10 -15 mol/min ⇒ 6.02 × 10 23 × 2 × 10 -15 molecule (O2)/min⇒
・ 12.04 × 10 8 molecules (O2)/min/cell (About 1.2 billion molecules of oxygen)
Even this is about 4 times the oxygen consumption calculated from the whole body.
● Red blood cells do not consume oxygen
Actually, out of the whole cell number 3.7 × 10 13 (37 trillion), 2.6 × 10 13, about 70 % is occupied by erythrocyte. From the common sense that the blood as a whole is 8 % of the body’s weight, although it is not convincing at first, it is that the results aggregated with the latest data are so. However, erythrocytes have a diameter of about 8 μm and are small as cells. (As measured by blood test, the number of erythrocytes is a value of about 5 million / μL, so if you calculate from now, 25 liters of erythrocytes will be included in the approximate blood volume of 5 liters per person.)
![]() Actually, erythrocytes do not have mitochondria, they are fairly specialized cells that procure ATP by glycolysis only. They do not use oxygen nor generate carbon dioxide. They do not use that oxygen despite having a lot. Priority is given to the important work that carries oxygen, although oxygen is full of them, they do not use oxygen themselves. Then in the case of a body weight of 60 kg, only about 10 trillion remaining cells will use oxygen.
Actually, erythrocytes do not have mitochondria, they are fairly specialized cells that procure ATP by glycolysis only. They do not use oxygen nor generate carbon dioxide. They do not use that oxygen despite having a lot. Priority is given to the important work that carries oxygen, although oxygen is full of them, they do not use oxygen themselves. Then in the case of a body weight of 60 kg, only about 10 trillion remaining cells will use oxygen.
Assuming that this number of cells supplements all of the ATP consumed per minute in the whole body as calculated from a metabolic rate of 1 day, the production of ATP per cell is divided by this cell number to 0.0694 × 7 / 6.6 × 10 -13 ⇒ 0.0736 × 10 -13 mol (ATP)/min/cell. The number of oxygen molecules required is 6/30 × 0.0736 × 10 -13 × 6.02 × 10 23 ⇒ 6 × 6.02 × 0.0736 / 30 × 10 10 ⇒ 0.0886 × 10 10 ⇒
· 8.86 × 10 8 molecule (O2) /min/cell (approximately 900 million molecular oxygen)
Approximately two thirds of the calculated value in cultured myocardial fibroblasts approached.
If there is no mistake in the above calculations, it seems that cultured cells are relatively expensive in oxygen consumption, but there are many cells with less oxygen consumption in the body. If it repeats, but there are no mistakes in calculation and way of thinking, please (check with yourself).
Oxygen consumption in one minute per cell calculated from the total metabolic rate for one day and the number of oxygen respiring cells 8.86 × 10 8 molecule (O2) /min/cell to consumption per second
0.147 × 10 8 molecule (O2) /s/cell (approximately 15 million molecular oxygen).
And, on the other hand, the cells discharge on average about 80% of the carbon dioxide of oxygen.
● Oxygen overflows from capillaries
Oxygen molecules are dissolved in 100 ml of water as a gas at 0.003 ml (3 × 10 -3 ml) per 1 mmHg of oxygen partial pressure. If it is 1 ml (1 cm3) of water, 0.00003 ml (3 × 10 -5 ml) is dissolved. In arterial blood, oxygen partial pressure is close to 100 mmHg, so 0.003 ml is dissolved per 1 ml of plasma. However, thanks to hemoglobin in the erythrocytes, the blood contains 0.204 ml of oxygen that is nearly 70 times its oxygen. When this comes to the peripheral capillary, the oxygen partial pressure falls to less than 40 mmHg at a stroke. Considering from the oxygen dissociation curve, at least about 30% of the oxygen contained in the hemoglobin is fully bursting out.
About 1 ml of blood, it is about 0.06 ml of oxygen molecules. However, only around 3 × 40 × 10 -5 ⇒ 1.2 × 10 -4 ml of oxygen per ml is dissolved in the surrounding tissue fluid. That is, it dissolves only the concentration of 0.2 % of the density in the blood. In skeletal muscle cells, oxygen may be absorbed by intracellular myoglobin. However, if it is already full of oxygen, of course, overflowing oxygen will be pushed thinly into the surrounding tissue.
If the cells in the middle do not consume oxygen and the oxygen partial pressure is still 40 mmHg, the oxygen overflowing from 1 ml of capillary blood will simply be calculated as 0.06 / 1.2 × 10 4 ⇒ 6 / 1.2 × 10 2 ⇒ 500 cm3. If the capacity of a cylindrical capillary is 1 ml, it is an image that covers a radius range of about 12 times its diameter. When the oxygen partial pressure is 1 mmHg, spread to the organization of 0.06 / 3 × 10 5 ⇒ 6000/3 ⇒ 2000 cm3. It corresponds to about 23 times the diameter of the diameter.
Since the diameter of the actual capillary is about 10 μm or less, it is about 200 μm in radius. Of course oxygen is consumed by surrounding cells, so the oxygen partial pressure drops sharply as it leaves the blood vessel, and the oxygen solubility decreases accordingly. Actually, it seems that cells are said to be within 200 μm radius around the capillary. It is surprising that there is not much difference in such a cumbersome calculation.
● How much surplus oxygen can afford
1 mol of gas is composed of 6.02 × 10 23 molecule and has a volume of 22.4 L (standard state). When the oxygen partial pressure is 1 mmHg, the oxygen molecules in the aqueous solution 1 cm3 are 3 × 10 -8 / 22.4 ⇒ 3 / 22.4 × 10 -8 mol (O2) / cm3. The number of molecules is 6.02 × 10 23 × 3 / 22.4 × 10 -8 molecule /cm3 ⇒ 6.02 × 3 / 22.4 × 10 15 molecule /cm3 ⇒ 18.06 / 22.4 × 10 15 molecule ⇒ 8.1 × 10 14 molecule (O2) /cm3.
On the other hand, the average oxygen consumption rate calculated from whole body cells is 0.147 × 10 8 molecule (O2) /s/cell (15 million molecules Of oxygen). Consider how much cells exist in the tissue mass 1 cm3. The number of 1 x 10 5 cells per 1 cm2 of the cultured cell sheet is simply diverted to a cube assuming each cell is a cube. Then, the number of cells is (√ (1 × 10 5)) 3 ⇒ 316 × 10 5 ⇒ 3.16 × 10 7 cell / cm3 (32 million cells). For this mass of tissue 1 cm3, the oxygen consumption per second is calculated as 4.64 × 10 14 molecule (O2) /s/cm3.
As a result, oxygen molecules that can dissolve in 1 ml of an aqueous solution with an oxygen partial pressure of 1 mmHg divided by the oxygen consumption rate of this tissue 1 cm3 yields 8.1 × 10 14 / 4.64 x 10 14 ⇒ 8.1 / 4.64 ⇒ It is consumed in 1.74 seconds.
 If this rough calculation is correct, if it thinks that it will disappear in a moment, 1 mmHg seems to have plenty of room. However, since oxygen released in the periphery should be in equilibrium where it is completely consumed in an instant, it may be that the lower oxygen partial pressure range is covered. Furthermore, if it is a cell that is metabolically active like the liver (because it consumes 16 times in this case), it can be guessed that the distance between the capillary and the cell must be much closer.
If this rough calculation is correct, if it thinks that it will disappear in a moment, 1 mmHg seems to have plenty of room. However, since oxygen released in the periphery should be in equilibrium where it is completely consumed in an instant, it may be that the lower oxygen partial pressure range is covered. Furthermore, if it is a cell that is metabolically active like the liver (because it consumes 16 times in this case), it can be guessed that the distance between the capillary and the cell must be much closer.
● Carbon dioxide gas that is discharged is also carried by the blood vessel
When the cell consumes oxygen, it will exhale it at least as much as 70% of its carbon dioxide. This is an empty shell after extracting all the energy that can be used biologically from the energy source sugar and fatty acid.It is also the final product of the citric acid cycle, and if this carbon dioxide gas is not tidied up, the citric acid cycle will not rotate normally even if there is oxygen. Even without worrying, carbon dioxide spreads from the cell to the capillary, contrary to oxygen, is absorbed by the blood flow returning to the heart and is recovered from the tissue. It is carried to the lungs by blood flow, and it is discharged from the mouth and nose. The respiratory movement of the lungs collectively exchanges the oxygen and carbon dioxide of cells in the body.
However, there is a condition that the concentration of carbon dioxide gas outside the body is sufficiently low to carry out carbon dioxide from the cell to the outside of the body. Carbon dioxide in breath exhaling normally is about 3 to 4 %, whereas the concentration of carbon dioxide in normal atmosphere is 0.03 % and roughly 100 times, so you can breathe in peace. Conversely, if the concentration of carbon dioxide in the inhaled air becomes 3 % or more, it will be impossible to expel carbon dioxide gas out of the body, so it is natural that there are times when the condition goes bad and it leads to death.
Although the unit is confusing, we estimated the gas exchange around the cell in a way that can be calculated even by junior high school students. Professionally, experts calculate using a frighteningly difficult function, but it is surprisingly deep and it can not be said that the clarity has not been clarified yet.
○ Referenced sites
・CELL BIOLOGY BY THE NUMBERS
・Adenosine triphosphate, Wikipedia, 28 June 2017
・On the number of human cells
An estimation of the number of cells in the human body, Annals of Human Biology, Bianconi et al., Annals of Human Biology
Volume 40, 2013.
・Oxygen consumption of cultured cells
Improvement of oxygen supply in hepatocyte culture, Yuki Sakai, Hepatocyte research group
・Oxygen consumption of the human heart cells in monolayer culture, Sekine, et al.(2014), (PubMed)
・Lack of Oxygen and Beyond(2009), http://bodyhacking.jp/anesth2009/
・Theoretical models of microvascular oxygen transport to tissue, Goldman(2008), (PubMed).
○ Related articles
◆[011] Body acid-base reaction and carbon dioxide gas ![]()
◆[023] Erythrocyte and hemoglobin ![]()
◆[016] Blood circulation ![]()
◆[029] Oxyhemoglobin dissociation curve ![]()
◆[010] Alveolar ventilation and dead space ![]()
◆[017] Absorption of carbohydratel ![]()
◆[047] Glycolysis ![]()
◆[042] TCA cycle ![]()
◆[019] Adenosine triphosphate ![]()
◆[046] Digestion and metabolism ![]()
◆[013] Lipid bilayer of the cell membrane ![]()
◆[007] Glucose and the hydrolysis of sucrose ![]()
◆[026] Interaction of myosin and actin ![]()
◆[015] Costal breathing ![]()
◆[038] Homeostasis of the core body temperature ![]()
○ Referenced books
・カラー版 ボロン ブールペープ 「生理学」, 西村書店
・プロッパー細胞生物学,化学同人
・Essential細胞生物学〈DVD付〉原書第3版,南江堂
・細胞の分子生物学, ニュートンプレス; 第5版 (2010/01)
・カラー図解 人体の正常構造と機能 全10巻縮刷版,坂井 建雄,日本医事新報社
・人体機能生理学,杉 晴夫,南江堂
・トートラ人体解剖生理学 原書8版,丸善
・柔道整復学校協会編「生理学」,南江堂
・東洋療法学校協会編「生理学」,医歯薬出版株式会社
rev.20160604,rev.20160619,rev.20161228,rev.20170506, rev.20170521, rev.20170709, rev.20180106, rev.20180109, rev.20180111.
KISO-IGAKU-KYOIKU-KENKYUKAI(KIKKEN)