[047] Glycolysis (GB#106A02) | 基礎医学教育研究会(KIKKEN)Lab

● Divide glucose into two pieces
Glucose that cells favorite is a mass of energy. Mitochondria transfer the majority of its energy to a number of adenosine triphosphate (ATP) that is easy to use. However, unexpectedly the glucose intact is not good, it does not fit the mitochondrial mouth. First of all, it is necessary to divide the glucose in half by “reaction of glycolysis”.
▼
—
Contents
- 1 ● Firewood splitting work of cells
- 2 ● Energy is slightly reduced after splitting
- 3 ● The missing energy has been transferred to other substances
- 4 ● The reaction of glycolysis is a bit confusing
- 5 ● Do not let glucose escape
- 6 ● Glucose is converted to fructose and phosphate group is added
- 7 ● Divided into two to become each fuel
- 8 ● Transfer the projecting energy to NADH and ATP
- 9 ● When another ATP is produced, the completion of pyruvic acid
- 10 ● After spending two ATPs, four are produced
- 11 ● Kaito-kei and Kaito
- 12 ○ Referenced sites
- 13 ○ Related articles
- 14 ○ Referenced books
● Firewood splitting work of cells
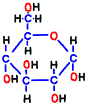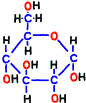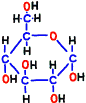
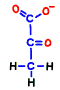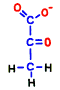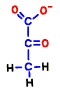
 Glucose is representative of human carbohydrates available. Basically, biological membranes themselves do not pass carbohydrates, as it required a special mechanism to take them into cells. Mitochondrial membranes are not as good as those of glucose. Glucose is divided in half to make two small molecules of organic acid called pyruvic acid. If mitochondria is a burning kiln that burns energy, it seems to break firewood into two pieces so that it can enter into the furnace mouth. However, one piece of pyruvic acid prepared by glycolysis is slightly smaller than half of glucose.
Glucose is representative of human carbohydrates available. Basically, biological membranes themselves do not pass carbohydrates, as it required a special mechanism to take them into cells. Mitochondrial membranes are not as good as those of glucose. Glucose is divided in half to make two small molecules of organic acid called pyruvic acid. If mitochondria is a burning kiln that burns energy, it seems to break firewood into two pieces so that it can enter into the furnace mouth. However, one piece of pyruvic acid prepared by glycolysis is slightly smaller than half of glucose.
● Energy is slightly reduced after splitting
Glucose is a compound with 6 carbon atoms, 1 mole (6.02 x 1023 molecules) has energy of 686 kcal. Whether it turns on fire or burning at once, it decomposes in the cell carefully, but when it finally becomes water and carbon dioxide, it releases this energy as a whole.
On the other hand, pyruvic acid has 3 carbons, 1 mole has energy of 282 kcal. In glycolysis, two molecules of pyruvic acid are produced from one molecule of glucose, but 2 moles of pyruvic acid causes only 564 kcal, twice that of 282 kcal. Deduction of energy of glucose and energy of two molecules of pyruvic acid, where did the 122 kcal go?
● The missing energy has been transferred to other substances
The advantage of glycolysis is that it creates two molecules of ATP at once, quickly, before the reaction of mitochondria. ![]() When mitochondria generates ATP, it finally needs oxygen molecule 02. But glycolysis does not need that oxygen. Also in the process of making pyruvic acid, ATP is made from adenosine diphosphate (ADP) in advance. Glycolysis is important as a production process of ATP without oxygen. However, the energy change of ADP → ATP is 7.3 kcal (value in the standard state) at 1 mol, and only 2 molecules is 14.6 kcal. In the actual intracellular environment, it will be a little bigger, but it will still be around 20 kcal.
When mitochondria generates ATP, it finally needs oxygen molecule 02. But glycolysis does not need that oxygen. Also in the process of making pyruvic acid, ATP is made from adenosine diphosphate (ADP) in advance. Glycolysis is important as a production process of ATP without oxygen. However, the energy change of ADP → ATP is 7.3 kcal (value in the standard state) at 1 mol, and only 2 molecules is 14.6 kcal. In the actual intracellular environment, it will be a little bigger, but it will still be around 20 kcal. ![]() Most of the difference energy is transferred to reduced form nicotinamide adenine dinucleotide (NADH), another type of energy carrier. This is energy that can not be transferred to ATP without the help of mitochondria, so it is usually not collected much attention when studying glycolysis, but the energy is being collected properly.
Most of the difference energy is transferred to reduced form nicotinamide adenine dinucleotide (NADH), another type of energy carrier. This is energy that can not be transferred to ATP without the help of mitochondria, so it is usually not collected much attention when studying glycolysis, but the energy is being collected properly.
● The reaction of glycolysis is a bit confusing
In short, glycolysis is a chemical reaction that transforms glucose into two pyruvic acids. In the process, this reaction produces two molecules of ATP and two molecules of NADH. That’s enough for beginner level physiology.
However, in medium textbooks, two molecules of ATP are used in the glycolysis reaction, and four molecules are made, so it is written that two molecules of ATP are deducted. In order to understand this it is necessary to look into the contents of the process a little more. The process is unexpectedly complicated, consisting of 10 steps of process, aligning with the TCA cycle which is apparently complicated and inaccessible. Roughly speaking, 5 stages in the first half are reactions that involve energy, and 5 stages later are responses to extract energy.
Every time, since exchange of each element of the reaction and exchange of energy are done by interaction with the enzyme, chemical reaction can not be accurately represented only by the compound as the substrate. With this, it is only necessary to grasp the approximate image.
● Do not let glucose escape
First of all, glucose entering the cell by the glucose transporter dedicated to cell membrane glucose remains out using the glucose carrier again if left as it is. Therefore, the enzyme that finds glucose first takes the phosphate group from ATP which is around there and adds it to glucose. 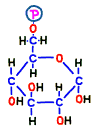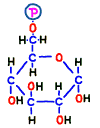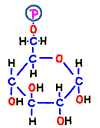 Then, it is no longer “glucose” but it is transformed into another substance called glucose 6-phosphoric acid (G6P). That is the reaction 1.
Then, it is no longer “glucose” but it is transformed into another substance called glucose 6-phosphoric acid (G6P). That is the reaction 1.
“6-Phosphoric acid” does not mean six phosphoric acids, as you can see from the structure. It means that phosphoric acid binds to the 6th carbon of the structure. Since there is no carriage that puts phosphoric acid stuck substances out like glucose-6-phosphate, it can not escape from the cell. And that energy of this phosphate may be thought of as a “spirit” for starting glycolysis reactions from now on.
(If there is sufficient ATP in the cell and there is not much need to replenish ATP by degrading glucose, replace the phosphate group with carbon number 1 in the structure. Then the glucose is turned towards the process of extending glycogen.)
● Glucose is converted to fructose and phosphate group is added
Next, prepare to add another phosphate group to glucose-6-phosphate. Later on, this process breaks down the compound and creates two identical molecules, so prepare another phosphoric acid beforehand so that it will not run away as soon as it is released.
Prior to that, in order to make the place (–CH2OH) to attach the phosphate group, in reaction 2, the annular structure of glucose is opened and the place of hydrogen is replaced. Elements that make up that do not change, but it has changed to another sugar called fructose. 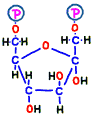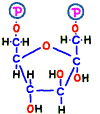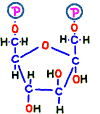 The compound is named fructose 6-phosphate.
The compound is named fructose 6-phosphate.
Immediately after that, fructose 1,6-bisphosphate is made by adding phosphate group from new ATP in reaction 3. Bisphosphate means two phosphates.
● Divided into two to become each fuel
In reaction 4 and 5, fructose 1,6-bisphosphoric acid is broken to finally produce two molecules of compound. Although this reaction is quite complicated, there is no entry particularly in terms of energy, so its explanation is omitted.
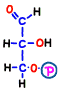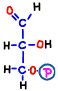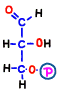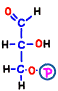 The finished 3-carbon sugar glyceraldehyde 3-phosphate is the substance at the end of the preparation period of the first half of glycolysis, and it spends two molecules of ATP per glucose molecule before coming here.
The finished 3-carbon sugar glyceraldehyde 3-phosphate is the substance at the end of the preparation period of the first half of glycolysis, and it spends two molecules of ATP per glucose molecule before coming here.
From now on, it’s the process of extracting more energy than this amount spent.
● Transfer the projecting energy to NADH and ATP
![]() First, in reaction 6, electrons and hydrogen having energy from glyceraldehyde 3-phosphate are absorbed by a reaction in which nicotinamide adenine dinucleotide (NAD+) becomes NADH.
First, in reaction 6, electrons and hydrogen having energy from glyceraldehyde 3-phosphate are absorbed by a reaction in which nicotinamide adenine dinucleotide (NAD+) becomes NADH. 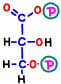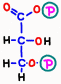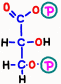 With the remaining energy, phosphoric acid that is staggering alone binds to 1,3-bisphosphoglyceric acid.
With the remaining energy, phosphoric acid that is staggering alone binds to 1,3-bisphosphoglyceric acid.
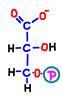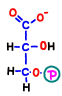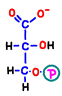 In reaction 7, the first ATP is generated by replacing the phosphate bond with its energy immediately with ADP. The remaining part turns into a substance called 3-phosphoglycerate (glycerate 3-phosphate).
In reaction 7, the first ATP is generated by replacing the phosphate bond with its energy immediately with ADP. The remaining part turns into a substance called 3-phosphoglycerate (glycerate 3-phosphate).
● When another ATP is produced, the completion of pyruvic acid
Even though one ATP can be made, one phosphate remains in this 3-carbon compound (glycerate 3-phosphate). This phosphate group is substituted for ADP in reactions 8, 9 and 10 to produce ATP. Reactions 8 and 9 have preprocessing to make ATP, but at this time, it does not matter. The substance from which the phosphate group has cleared is at last pyruvic acid.  There is still 282 kcal of energy remaining in pyruvic acid. The majority of the 61 kcal, which is 282 kcal minus the half value of glucose energy, was transferred to NADH and ATP, and a small portion that was not transferred worked as the driving force to advance the whole reaction.
There is still 282 kcal of energy remaining in pyruvic acid. The majority of the 61 kcal, which is 282 kcal minus the half value of glucose energy, was transferred to NADH and ATP, and a small portion that was not transferred worked as the driving force to advance the whole reaction.
![]()
![]()
● After spending two ATPs, four are produced
After all, in the first half, before disassembling one molecule of glucose which is a hexose sugar into two, first decompose two molecules of ATP and attach two phosphate groups therefrom to make two identical three monosaccharides . After that, each process extracts a little energy of 3 monosaccharides, each producing 2 molecules of ATP, pyruvic acid is made. Since 2 molecules x 2 = 4 molecules of ATP are recovered later by consuming two molecules of ATP, two molecules of ATP are collected from one molecule of glucose. Also remember that there are two molecules of NADH. It is estimated that this enters mitochondria and produces three molecules of ATP. Of course, at this time pyruvic acid also enters the mitochondria, and the TCA circuit sucks up the energy of the drone, eventually producing approximately 30 molecules of ATP in all.
However, it is only condition that NADH is processed and ATP is born only oxygen breathing can be done. If the cell’s oxygen molecule O2 is insufficient, mitochondria will be closed and only 2 molecules of ATP made directly in the glycolysis can be increased. 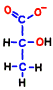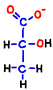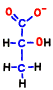 At that time, NADH which does not enter mitochondria presses 2H+ against pyruvic acid, converts it to stable lactic acid, and itself becomes NAD+. That’s why lactic acid increases in anoxic breathing.
At that time, NADH which does not enter mitochondria presses 2H+ against pyruvic acid, converts it to stable lactic acid, and itself becomes NAD+. That’s why lactic acid increases in anoxic breathing.
● Kaito-kei and Kaito
In Japan, it is ambiguous to say “Kaito-kei” or “Kaito” by textbook as a word that describes the process by which glucose to pyruvate, possibly lactate, can be formed. Which is right is that neither is correct, either. There are explanations that so-called nuances are somewhat different, but it also varies, and both “Kaito-kei” and “Kaito” are simply glycolysis in English, so there is no such distinction internationally. In technical terms, -kei is usually equivalent to -system in English, I guess they are using it because it’s a little cool.
○ Referenced sites
・解糖系という概念に対する異論, 歴史生物学への誘い, 2014年1月8日.
・炭水化物とアルコール, 生活習慣病を予防する食生活, 佐藤章夫.
○ Related articles
◆[007] ブドウ糖と,ショ糖の加水分解 glucose and the hydrolysis of sucrose ![]()
◆[017] 糖質の吸収 absorption of carbohydratel ![]()
◆[050] Cellular respiration ![]()
◆[027] ペプチド結合 peptide bond ![]()
◆[032] グリコーゲンの合成 glycogen synthesis ![]()
◆[042] TCA回路 TCA cycle ![]()
◆[019] アデノシン三リン酸(ATP) adenosine triphosphate ![]()
◆[026] Interaction of myosin and actin ![]()
◆[011] 体内の酸性・アルカリ性と炭酸ガス body acid-base reaction and carbon dioxide gas ![]()
○ Referenced books
・Lehninger Principles of Biochemistry 6th, International Edition, Macmillan Higher Education, England.
・Essential細胞生物学〈DVD付〉原書第3版,南江堂
・カラー版 ボロン ブールペープ 「生理学」, 西村書店
・カラー図解 人体の正常構造と機能 全10巻縮刷版,坂井 建雄,日本医事新報社
・トートラ人体解剖生理学 原書8版,丸善
・柔道整復学校協会編「生理学」,南江堂
・東洋療法学校協会編「生理学」,医歯薬出版株式会社
rev.20150202,rev.20150315,rev.20160507,rev,20160618, rev.20170211,rev.20170506,rev.20170709.
KISO-IGAKU-KYOIKU-KENKYUKAI(KIKKEN)