[032] Glycogen synthesis (GB#101D02) | 基礎医学教育研究会(KIKKEN)Lab

● When the same thing connects it becomes another thing
Glycogen is a special form for storing large amounts of glucose in cells. Thousands, tens of thousands of glucose are connected in tufts to become one macromolecule. Especially in the liver and skeletal muscle cells, storage of a large amount of glucose is necessary. It seems that “number” of molecules is more problematic than size in cells. When cells are used, it is necessary to separate glucose, but it is inevitable to hold the same molecule in large quantities separately. When tens of thousands of molecules are joined into one, the “number” of molecules becomes one tens of thousands. It is a different substance between 10,000 glucose and 1 glycogen. It is well done.
—
Contents
● Small handwork
Although tens of thousands of glucose are connected, the work of synthesizing glycogen is very detailed. There is no other choice but to keep glucose one step further. When making proteins, every single amino acid sequence is important, so everyone will be amazed at the precision of the mechanism that keeps it one by one. On the other hand, glycogen is a polymer of the same glucose alone. I guess that you ought to quickly coalesce, but cells can not do that. As necessary, glucose is inherited, detached and adjusted, so it may be convenient to take one by one. Glycogen always has an edge where glucose is added and released. Moreover, it is structured that it is countless at the same time.
● Energy required for connection
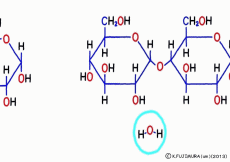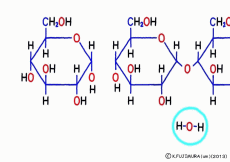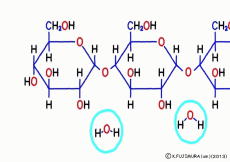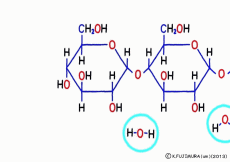
Just mixing glucose in water does not make glycogen. When glucose is inherited to glycogen, as in the case of making sucrose, and in the same way as when making proteins from amino acids, simply connect with “dehydration condensation”. So, a certain amount of energy is required for this connection. Speaking of the source of energy for cellular reactions, ATP (adenosine triphosphate) is the mainstream nucleotide. However, what works directly in glycogen synthesis is another nucleotide called UTP (uridine triphosphate).![]() Even though it says another, adenosine is replaced by a base called uridine, and three phosphates are connected, so that it works the same as a high energy source.
Even though it says another, adenosine is replaced by a base called uridine, and three phosphates are connected, so that it works the same as a high energy source.
● The direction to connect is fixed
The basic form of glycogen is repetition of glucosidic bond (glycosidic bond) in which glucose (in accordance with regulation number) No.1 and the carbon atom No.4 of the neighboring glucose are linked by oxygen atoms as a result of dehydration condensation. If you put the chain sideways, it looks like the same way if you extend to the right or extend to the left. However, as the work of the cell, since the working enzyme is stubborn, the direction to extend is only one that connects No. 1 of new glucose to the 4th end of the chain. (Actually, although it branches many times on the way, this is done by another enzyme in a special way.)
● What need before consolidating
Glucose flowing outside the cell is caught by a cellular membrane glucose transporter and incorporated into the cell. Upon entering the cell, an enzyme called glucokinase will immediately turn glucose into glucose 6-phosphate (G6P) by coupling phosphoric acid to carbon No.6. Carrier protein is like a revolving door dedicated to glucose, it is also an entrance as well as an exit for naked glucose. Therefore, as it is, glucose taken in is escaping to the outside. On the other hand, there is no revolving door through which G6P can pass through the cell membrane. That is why phosphoric acid is attached to it, although it uses one ATP, it is an important procedure to make the captured glucose impossible to pass through the revolving door. However, if G6P remains unchanged, it will be caught up by the glycolysis system and disassembled this time to get rid of energy. When linking to glycogen, replace this phosphoric acid with carbon No.1 and convert it to glucose monophosphate (G1P).![]() This reaction can proceed with an enzyme called glucose phosphomutase, but because it just replaces the phosphate group, there is no energy consumption for deduction. The linking work to glycogen starts here.
This reaction can proceed with an enzyme called glucose phosphomutase, but because it just replaces the phosphate group, there is no energy consumption for deduction. The linking work to glycogen starts here.
● UTP once delicious twice
 A high energy substance called UTP works for synthesis of glycogen. However, it is not directly connected with glycogen by glucose directly in one shot, it is a little complicated mechanism.
A high energy substance called UTP works for synthesis of glycogen. However, it is not directly connected with glycogen by glucose directly in one shot, it is a little complicated mechanism.
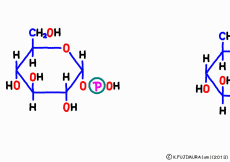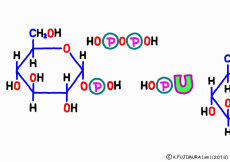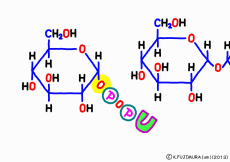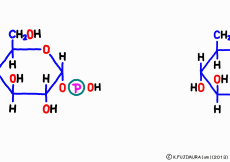
First, uridine triphosphate is cleaved from diphosphate (pyrophosphate) by the enzyme,![]() and once the form of uridine monophosphate (UMP) is formed, it is linked to the phosphate group of G1P. It seems to boost this reaction by separating pyrophosphoric acid which immediately hydrolyzes and provides energy. As a result, a form in which uridine diphosphate (UDP) sticks to glucose is completed, which is called UDP-glucose.
and once the form of uridine monophosphate (UMP) is formed, it is linked to the phosphate group of G1P. It seems to boost this reaction by separating pyrophosphoric acid which immediately hydrolyzes and provides energy. As a result, a form in which uridine diphosphate (UDP) sticks to glucose is completed, which is called UDP-glucose.
UDP When glucose is reached, it finally enters the linking process. Captured by a new enzyme called glycogen synthase, the portion of UDP just freshly caught is stripped off, and the naked glucose is connected to the origin of glycogen originally at one end. Tear-off pyrophosphoric acid also draws out energy, so uridine triphosphate (UTP) seems to have absorbed drastic energy. But in terms of income and expenditure, in order to link one glucose, UTP is the only way UDP can be formed by losing one phosphoric acid.
● Branching, increasing efficiency
Glycogen is not single-stranded, actually it branches off when 8 to 12 glucose is connected, it has been repeated over and over again. Unlike simply linking glucose, the mechanism of branching is longer to explain here, so it will be another opportunity. However, by branching, there is an advantage that many glucose can be connected to glycogen at the same time in each end of countless branches, even in a stabbing operation in each case. This is the same when cutting out glucose as necessary. Although how to synthesize glycogen and how to decompose are completely different, even when decomposing, glucose must be cut out one by one. At that time too, if there are several branches, you can cut out a lot of glucose from the end of each branch all at once.
● The beginning of glycogen is not glucose
Glycogen is actually not a mass of pure glucose. When making a new glycogen, will it first link glucose to glucose? Then it is a disaccharide called maltose. And if one dextrose is connected, a trisaccharide called maltotriose is produced. However, in cellular machinery, such naked oligosaccharides can not be used as glycogen seeds. The beginning of glycogen is not a sugar but a special protein with a strange name called glycogenin. Glycogenin is an enzyme that has two glucose linking moieties in opposite directions to each other, and acts as a glycogen primer. Then link themselves with the first UDP glucose and the following three to four UDP glucose. Glycogen is still fundamentally attached even when glycogen grows, so glycogen is somewhat different from a simple polymer of glucose.
Note:
· ΔG of hydrolysis of glycoside bond of glycogen is -15.5 kJ/mol (-3.7 kcal/mol)
· ΔG of hydrolysis of pyrophosphoric acid is -33.5 kJ/mol (-8.0 kcal/mol)
・For more information on glycogenin(wikipedia)
○ Referenced sites
・Biochemistry of Metabolism,Instructional materials for a studio-format course(Rensselaer Polytechnic Institute (RPI)) (www.rpi.edu/dept/) ← link broken(2017-07-25)
・Glycogen/Biochemistry 200/Stanford University School of Medicine
○ Related articles
◆[007] Glucose and the hydrolysis of sucrose ![]()
◆[017] 糖質の吸収 absorption of carbohydratel ![]()
◆[027] ペプチド結合 peptide bond ![]()
◆[019] アデノシン三リン酸(ATP) adenosine triphosphate ![]()
◆[047] Glycolysis ![]()
◆[042] TCA回路 TCA cycle ![]()
◆[050] Cellular respiration ![]()
◆[006] Osmotic pressure ![]()
◆[037] 膠質浸透圧 colloid osmotic pressure ![]()
◆[046] Digestion and metabolism ![]()
○ Referenced books
・トコトンわかる図解 基礎生化学,池田和正,オーム社
・プロッパー細胞生物学: 細胞の基本原理を学ぶ,化学同人
・Essential細胞生物学〈DVD付〉原書第3版,南江堂
・細胞の分子生物学, ニュートンプレス; 第5版 (2010/01)
・肉単―ギリシャ語・ラテン語 (語源から覚える解剖学英単語集 (筋肉編))
・カラー図解 人体の正常構造と機能 全10巻縮刷版,坂井 建雄,日本医事新報社
・人体機能生理学,杉 晴夫,南江堂
・トートラ人体解剖生理学 原書8版,丸善
・イラスト解剖学,松村 讓兒,中外医学社
・柔道整復学校協会編「生理学」,南江堂
・東洋療法学校協会編「生理学」,医歯薬出版株式会社
rev.20150504, rev.20150505, rev.20160204,rev.20160612, rev.20170505, rev.20180408.
KISO-IGAKU-KYOIKU-KENKYUKAI(KIKKEN)